BY LETTER
Carbon Nanotube
Technology > Application > Construction
Technology > Application > Materials Technology
Science > Chemistry > Organic Chemistry
Technology > Application > Materials Technology
Science > Chemistry > Organic Chemistry
Buckytube, Nanotube, Carbon Nanofibre | |
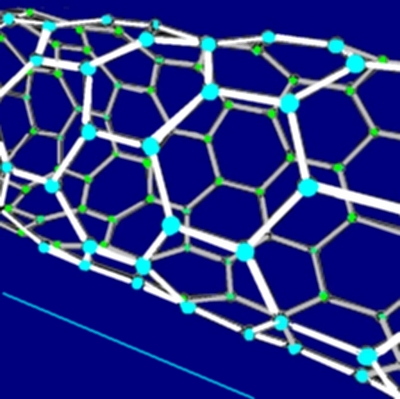 Image from Steve Bowers | |
| Carbon Nanotube molecule, with a much smaller magcarbon nanotube alongside for comparison. | |
Elongated fullerene carbon molecule in a tubular configuration. Nanotubes are cylinders arranged from a single layer of carbon atoms. They are are 10 times stronger than, and have a fraction of the weight of, steel. The self-repairing variant Polyfullene is particularly useful.
As well as being strong they are also heat resistant, formed from the same hexagonal patterns as graphene, arranged in three different ways; armchair, zigzag and chiral (each with a different diameter and bulk density).
An essential part of of nanotech and nanofacturing, nanotubes can perform the same electronic functions of silicon-based components, but at a much finer scale. Nanotubes are standard in mesoscale molecular computer chips because their electrical conductivity can be controlled by the extent they are twisted. During the middle information period, nanotubes enabled the continued exponential growth of computing and integrated circuits that was referred to as Moore's Law, as well as allowing the creation of three-dimensional circuits.
Other uses for nanotubes include spacecraft construction, beanstalk cables and other forms of space tether, and as reinforcement in rotation space habitats such as Bishop Rings and McKendree Cylinders. Because of their many useful properties, and the relative ease of construction, nanotubes are still manufactured in bulk, even in relative primitive polities and societies.
Related Articles
- Adamant
- Beanstalk
- Bishop Ring
- Buckyball - Text by M. Alan Kazlev
Short for buckminsterfullerene, and part of a larger class of molecules known as as fullerenes, the third form of carbon. Any of a number of roughly spherical molecule formed of a large number of carbon atoms. The smallest and most common form of buckminsterfullerene is C60; it has 60 carbon atoms in a soccer-ball shape, but there are many variants. The third form of carbon. After atomic age visionary thinker and inventer R. Buckminster Fuller's building designs. - Carbon
- Diamondoid
- Fullerene
- Fullerite - Text by M. Alan Kazlev
Atomically precise fullerene-like material, contains magmatter elements that greatly enhance performance. Widely used by some ISOs. - McKendree Cylinder
- Pandifico: Elastic Diamondoid Fiber Composites
- Technology Timeline
Appears in Topics
Development Notes
Text by M. Alan Kazlev
Initially published on 09 December 2001.
Initially published on 09 December 2001.